|
The saturation vapor pressure of water at any given temperature is the vapor pressure when relative humidity is 100%. The vapor pressure of water may be calculated from the saturation vapor pressure and relative humidity. R v = Specific gas constant for water vapor, 461.495 J/(kg R d = Specific gas constant for dry air, 287.05 J/(kg Using this method, error in the density calculation is less than 0.2% in the range of −10 ☌ to 50 ☌. In this case, the partial pressure of water vapor is known as the vapor pressure. The density of humid air may be calculated as a mixture of ideal gases. Hence the mass per unit volume of the gas (its density) decreases. So when water molecules (vapor) are introduced to the air, the number of air molecules must reduce by the same number in a given volume, without the pressure or temperature increasing. For any gas, at a given temperature and pressure, the number of molecules present is constant for a particular volume. This occurs because the molecular mass of water (18) is less than the molecular mass of air (around 29). The addition of water vapor to air (making the air humid) reduces the density of the air, which may at first appear contrary to logic. At standard ambient temperature and pressure (70 ☏ and 14.696 psia), dry air has a density of ρ SATP = 0.075 lb m/ft 3 ~ 1.2 kg/m 3.At standard ambient temperature and pressure (25 ☌ and 100 kPa), dry air has a density of ρ SATP = 1.168 kg/m 3.At standard temperature and pressure (0 ☌ and 101.325 k Pa), dry air has a density of ρ STP = 1.292 kg/m 3.The specific gas constant R for dry air is: 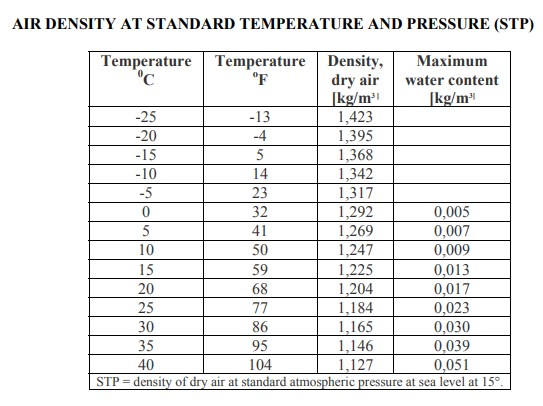
Where ρ is the air density, p is pressure, R is the specific gas constant, and T is temperature in kelvins. So here, if we were doing in terms of six figs, that comes out to 22 times 10 to the six kg.The formula for the density of dry air is given by: And that's gonna give us the best answer. So ideally we want to do it in terms of one sig fig. Now here we want it in terms of sick fix. So that comes out to be 1.77, 6 times 10 to the six kg. And then finally one kg is 10 to the six g or to the three g. And then one microgram is equal to 10 to the -6 g. Remember centimeters cubed is the same thing as Ml So we're gonna have. So again it comes from multiplying this number with this number Now that I have centimeters cubed. Now when we multiply these centimeters squared by this centimeter, multiplying them together gives me centimeters cubed that comes out to be 5.9207 times 10 to the 15 centimeters cubed. So remember one ft is 12", 1" is 2.54 cm. Next I would take my 15 ft and I'd convert that also into cm. This comes out to 1.294 9, 9 times 10 to the 13 cm squared. And then we'd say finally that one cm is equal to 10 to the -2 m. And then we're gonna stay here, one km is 10 to the three m. And the conversion factor is that one kilometer is equal 2.621371 miles here I squared so that the mile square cancel out. So we're gonna start out with our 500 miles squared. So that's a clear giveaway that I need to convert miles squared and feet both into centimeters and then multiply them together to get centimeters cubed. Remember that centimeters cubed, one centimeters cubed is equal to one millimeter. Our density is in micrograms per mil leader. Alright, so here, what we need to do first is we need to basically get the units to match up. 
So here it says a lake with a surface area of 500 miles squared and an average depth of 15 ft is polluted with 150.3 micrograms of mercury per millimeter of mercury, calculate the total mass in kilograms of mercury in the lake.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
